Journal Article in Applied System Innovation:
This work highlights the potential of wearable technologoies to monitor muscle activity changes during stroke recovery in acute clinical settings and their importance for motivation and understanding of progression from the survivor’s point of view: ‘I was hopeful that it would show signs of things that are occurring when I couldn’t physically feel it…if you had other scientific evidence that things were happening, even beyond their notion that it would, it gives you a lot of hope. You just have to be patient, and it’s harder to take when someone tells you, but easier to understand if someone actually shows you’.
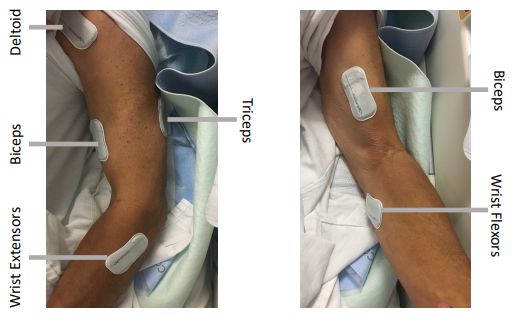
Aim: Describe the use of wireless sEMG sensors to examine changes in muscle activity during acute and subacute phases of stroke recovery, and understand the participant’s perceptions of sEMG monitoring.
Method: Muscle activity was tracked by five wireless sEMG sensors beginning three days post-stroke and continued through discharge from inpatient rehabilitation. Activity logs were completed each session, and a semi-structured interview occurred at the final session with three- and eight-month follow-up sessions.
Results: The longitudinal monitoring of muscle and movement recovery in the clinic and community was feasible using sEMG sensors. The participant and medical team felt monitoring was unobtrusive, interesting, and motivating for recovery, but desired greater in-session feedback to inform rehabilitation.
Interpretation: This work highlights that barriers in equipment and signal quality still exist, but capitalizing on wearable sensing technology in the clinic holds promise for enabling personalized stroke recovery.


 The Ability & Innovation Lab is excited to announce that two of our recent grant proposals have been funded! This funding will help to accelerate our mission to improve movement for individuals with neurologic disorders.
The Ability & Innovation Lab is excited to announce that two of our recent grant proposals have been funded! This funding will help to accelerate our mission to improve movement for individuals with neurologic disorders.